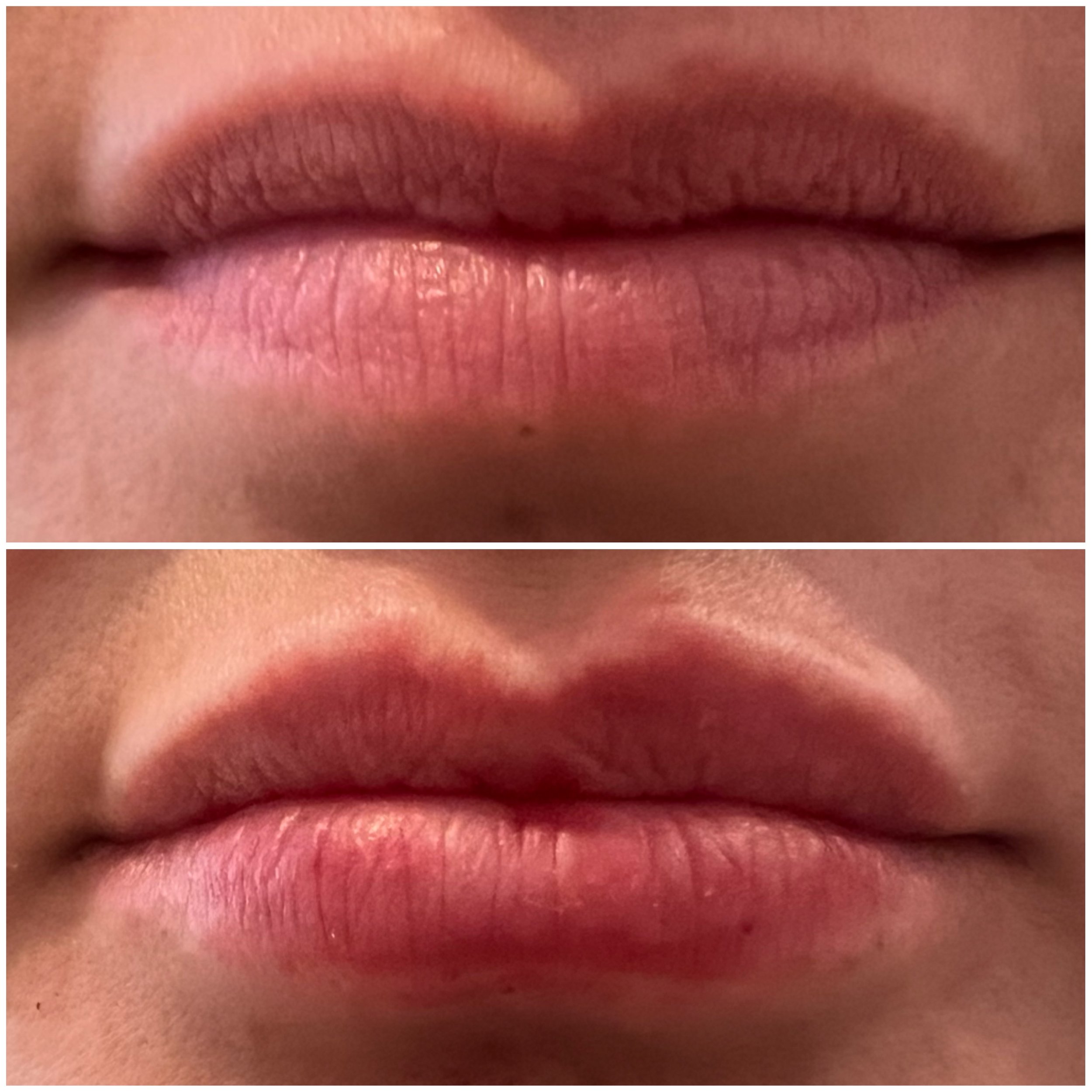
The ART & SCIENCE of SKIN and BODY TRANSFORMATION
“At Sterling Aesthetics, our mission is to provide a wholistic and personalized approach to restorative skincare by REVERSING AND SLOWING THE SIDE EFFECTS that can come with the aging process and environmental stressors. Based on ones skincare concerns, general health and lifestyle, At SA we can help you achieve your skincare and aesthetic goals.” - Dr. K